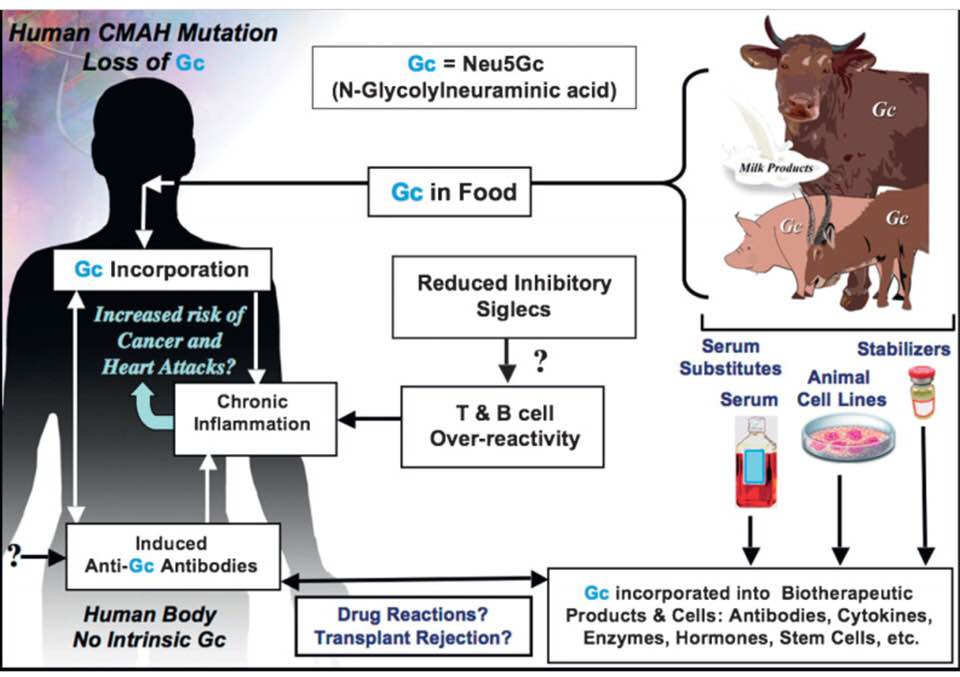
Sialic acids are monosaccharides (sugars) typically found at the outer ends of glycan chains (glycoconjugates aka glycocalyx) covering the surface of all vertebrate cells. The most commonly expressed sialic acid is N-acetylneuraminic acid (Neu5Ac), which is the precursor for N-glycolylneuraminic acid (Neu5Gc) synthesis, via action of the CMP-N-acetylneuraminic acid hydroxylase (CMAH) enzyme. Humans lost the ability to make Neu5Gc at the time our species diverged from chimps & gorillas 8 (2-3 in another paper) million years ago. Instead humans make Neu5Ac, as do elephants, chickens & shellfish.
Although Neu5Gc is foreign to humans, our intracellular biochemical pathways cannot distinguish between Neu5Ac and Neu5Gc, which differ by one oxygen atom. Thus, exogenous Neu5Gc is taken up and metabolically incorporated into human cells, eventually being expressed on the cell surface as if it were made in the same cell. Furthermore, orally ingested Neu5Gc is taken up into the human body, & small amounts of Neu5Gc are present in normal human tissues. Interestingly, the Neu5Gc-rich foods identified to date are red meats, which have been associated with circulating inflammatory markers indicating endothelial dysfunction.
Although Neu5Ac and Neu5Gc seem biochemically indistinguishable to human intracellular biosynthetic pathways, the latter is immunogenic in humans, leading to anti-Neu5Gc–specific antibody responses. Although such antibodies had been reported in some diseases, it was only recently recognized that all adult humans express them. Indeed, normal humans can express high titers of circulating anti-Neu5Gc immunoglobulin IgG & IgM antibodies against a variety of Neu5Gc-containing epitopes commonly found on endothelial cells.
These data indicate a novel human-specific mechanism in which Neu5Gc-rich foods deliver immunogenic Neu5Gc to the endothelium, giving anti-Neu5Gc antibody- and complement-dependent activation, and potentially contributing to human vascular pathologies. In the case of atherosclerosis, Neu5Gc is present both in endothelium overlying plaques and in subendothelial regions, providing multiple pathways for accelerating inflammation in this disease.
For further details, click the following link:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2792214/
 RSS 訂閱
RSS 訂閱
